News Story
NIH Funds "Smart Pill" for Gut Health Monitoring

University of Maryland (UMD) researchers will continue to advance their “smart pill” that tracks gut health, with support from a National Institutes of Health (NIH) R21 grant. Distinguished University Professor Reza Ghodssi (ECE/ISR/Fischell Institute) is the Principal Investigator (PI) and Younggeon Jin (Animal & Avian Sciences), Justin Stine (MATRIX/ENGR), and Pankaj Pasricha (Mayo Clinic) are co-PIs.
The work, which began at the University of Maryland, College Park campus, will now extend to Southern Maryland, into the UMD MATRIX Lab.
“This will greatly expand the research scope of our ingestible capsule work. We are taking a systems approach to improving the reliability of ingestible sensors beyond what we previously thought was possible,” Dr. Ghodssi said. “Establishing trusted measurements is essential to unlocking the next generation of autonomous, closed-loop technologies for the GI tract. We thank the National Institutes of Health for supporting this life-changing work.”
Earlier, Less Invasive Detection
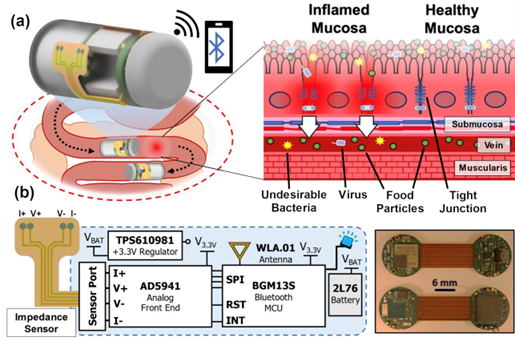
The pill will support those with Crohn's disease and ulcerative colitis, collectively known as inflammatory bowel disease (IBD). IBD causes inflammation in the gastrointestinal (GI) tract, but because inflammation is a common symptom of other disorders, diagnosis is complicated. Capsule endoscopy is minimally invasive tool that provides images of the GI tract to simplify diagnosis for patients but doesn’t provide health measurements and is only used reactively, after symptoms appear.
One way to gather specific data and enable early-stage assessment of intestinal tissues is using capsule platforms to detect bioimpedance, a measure of how well the body resists electric current flow. Inflamed tissue is more permeable and often has more fluid, disrupted cell barriers, and changes in ion movement. These factors impact electric conductivity, making bioimpedance a promising technique for quantifying inflammation.
Researchers have established the potential of bioimpedance for inflammation assessment and monitoring in their previous work, however, studies of bioimpedance beyond the esophagus remain limited. This is in part because measurements don’t behave the same everywhere or for everyone, which makes interpretation harder. Another hurdle is inconsistent sensor contact, which obscures real-time correlations between impedance and inflammation.
Advancing and Improving the Capsule
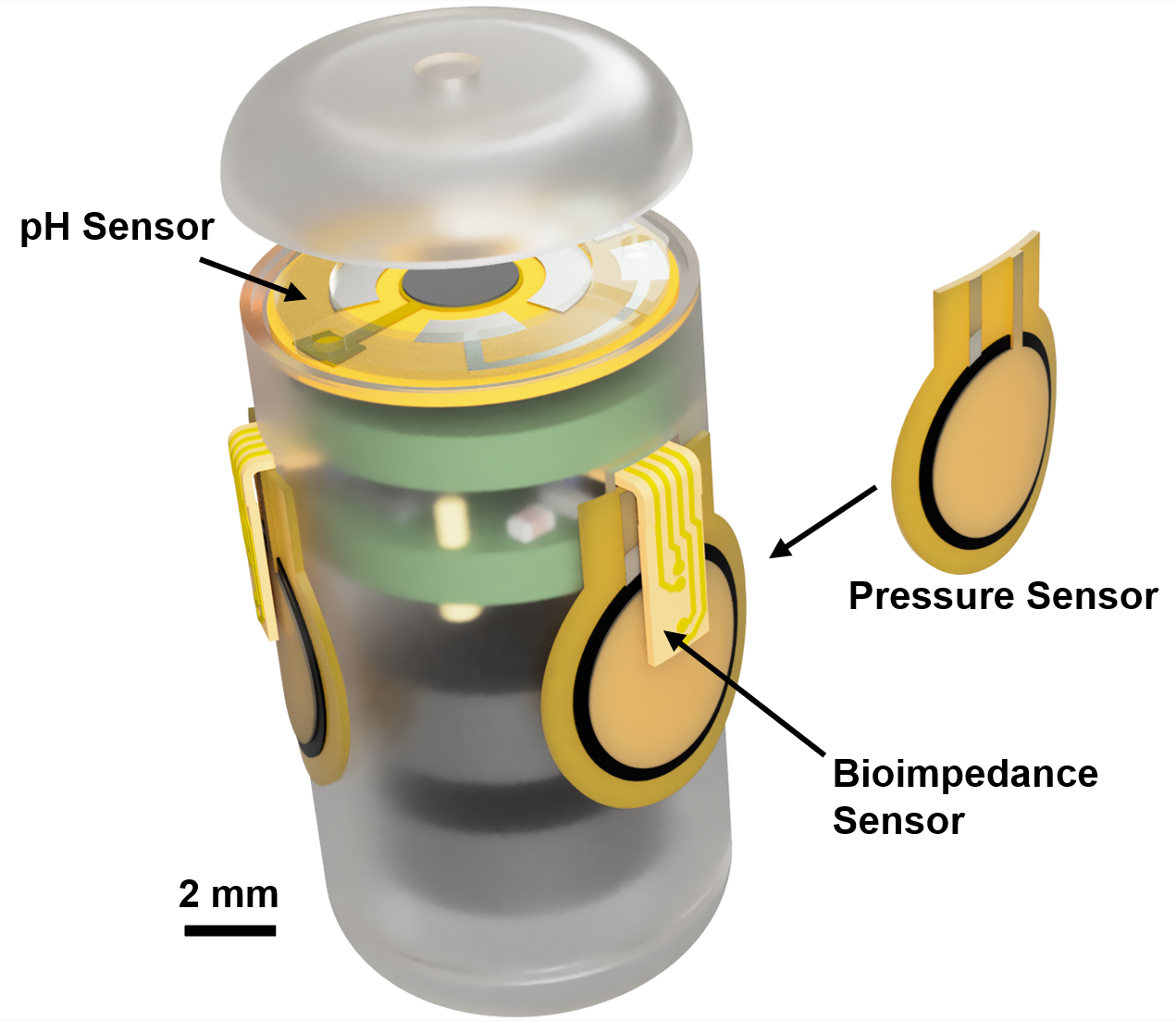 The NIH R21 grant will help improve the capsule with mechanisms to detect and verify good sensor contact so researchers can identify reliable data. The MATRIX’s Advanced Manufacturing Lab (AML @ SMART) is fully equipped for supporting this work. It will be used for developing sensors and capsule packaging, integrating the sensors into the system, and evaluating the capsule prototype. Co-PI Dr. Stine is the lab’s Director of Remote Sensing and Microsystems.
The NIH R21 grant will help improve the capsule with mechanisms to detect and verify good sensor contact so researchers can identify reliable data. The MATRIX’s Advanced Manufacturing Lab (AML @ SMART) is fully equipped for supporting this work. It will be used for developing sensors and capsule packaging, integrating the sensors into the system, and evaluating the capsule prototype. Co-PI Dr. Stine is the lab’s Director of Remote Sensing and Microsystems.
“The GI tract presents a challenging sensing environment; we employ biofeedback and multimodal sensing to account for measurement variability. We look forward to advancing this work with the team and realizing a viable prototype,” said Dr. Stine.
This new iteration is expected to expand capabilities for proactive disease monitoring and personalized treatment optimization, ultimately improving long-term patient outcomes. The work includes two future aims: Building and validating the sensing system in controlled environments and testing the system’s safety and real-world performance inside living organisms. The capsule is in development and not currently available to the medical community.
Published March 31, 2026